The Science of Bioreactors: Key Insights and Advanced Techniques
Bioreactors sit at the intersection of biology and engineering, providing carefully controlled environments where cells, enzymes, or microorganisms can grow and produce valuable products. From vaccines and antibodies to biofuels and food ingredients, these systems enable large-scale, consistent, and safe production that would be impossible in simple flasks or small laboratory vessels.

From vaccines to food enzymes, many products depend on keeping living cells productive inside a controlled vessel. A bioreactor is that vessel plus the hardware and software needed to regulate temperature, mixing, oxygen, nutrients, and sterility. The science is less about a single tank design and more about managing the biological “factory” so it behaves predictably as conditions, volumes, and production targets change.
Bioreactor technology: design choices that matter
Bioreactor technology starts with selecting a configuration that matches the biology and the process goal. Stirred-tank reactors are common because they scale well and can be instrumented extensively, but shear-sensitive cultures (such as some mammalian cells) may require gentler mixing strategies, specialized impellers, or alternative reactor types. Single-use bioreactors reduce cleaning validation and turnaround time, while stainless-steel systems remain common for very large volumes, long campaigns, or processes requiring aggressive cleaning.
Key design parameters include geometry (height-to-diameter ratio), impeller type, sparger design, and heat-transfer surface area. These directly influence mixing time, gas–liquid mass transfer, and temperature control. For aerobic processes, oxygen transfer is often the limiting factor; engineers focus on improving the volumetric mass-transfer coefficient while avoiding foam formation and excessive shear. For anaerobic processes, exclusion of oxygen, tight control of redox conditions, and safe handling of off-gases become more central.
Instrumentation is part of the “technology” as much as the tank itself. Modern systems typically integrate pH, dissolved oxygen, temperature, and pressure sensors, with increasing use of capacitance probes (for viable biomass proxies), Raman or near-infrared spectroscopy (for in-line chemistry), and soft sensors that infer unmeasured variables from process data.
Bioprocessing systems: monitoring, control, and automation
Bioprocessing systems combine the reactor, upstream media preparation, gas supply, pumps, filtration, and digital control layers into one coordinated workflow. The difference between “running a culture” and operating an industrial process is the rigor of control: small disturbances that are harmless at bench scale can cause large productivity losses at scale.
Control strategies commonly include cascades, such as adjusting agitation and oxygen enrichment to maintain a dissolved oxygen setpoint. Advanced setups use model-based control or adaptive control to respond to changing oxygen demand, viscosity, or cell density over time. Batch, fed-batch, and continuous modes each impose different control priorities. Fed-batch requires precise feed-rate control to avoid overflow metabolism in microbes or to manage osmolality in cell culture. Continuous processing adds the challenge of maintaining steady states and preventing contamination over longer durations.
Automation and data integrity are increasingly important. Even outside regulated pharmaceutical settings, consistent electronic batch records, alarm management, and sensor calibration routines reduce variability. A practical “advanced technique” in many plants is designing a robust sampling plan: pairing in-line analytics with fewer, higher-value off-line assays (such as metabolite panels, cell viability, or product titer) to minimize contamination risk and operator-to-operator variation.
Industrial fermentation: scaling microbes without losing yield
Industrial fermentation typically uses bacteria, yeast, or fungi to produce biomass, metabolites, or recombinant products. Scaling these systems is not a simple matter of multiplying volumes, because mixing and oxygen transfer do not scale linearly. Large tanks can develop gradients in dissolved oxygen, pH, substrate concentration, and temperature. Microbes passing through these micro-environments may switch metabolic states, affecting yield, byproduct formation, and stress responses.
Advanced scale-up work often centers on selecting scale criteria that reflect what the organism “feels.” Depending on the process, teams may prioritize constant power input per volume, constant tip speed, or constant oxygen transfer capacity. None is universally correct; the right criterion depends on whether the main risk is oxygen limitation, shear sensitivity, foaming, or heat removal.
A related technique is scale-down modeling, where a lab or pilot system deliberately reproduces large-scale gradients (for example, by oscillating cells between well-mixed and oxygen-limited zones). This helps identify strains or feeding strategies that are resilient to industrial realities. Another key operational tool is antifoam management: excessive antifoam can reduce oxygen transfer and complicate downstream processing, so processes may combine mechanical foam breakers, optimized sparging, and minimal antifoam dosing based on foam sensors.
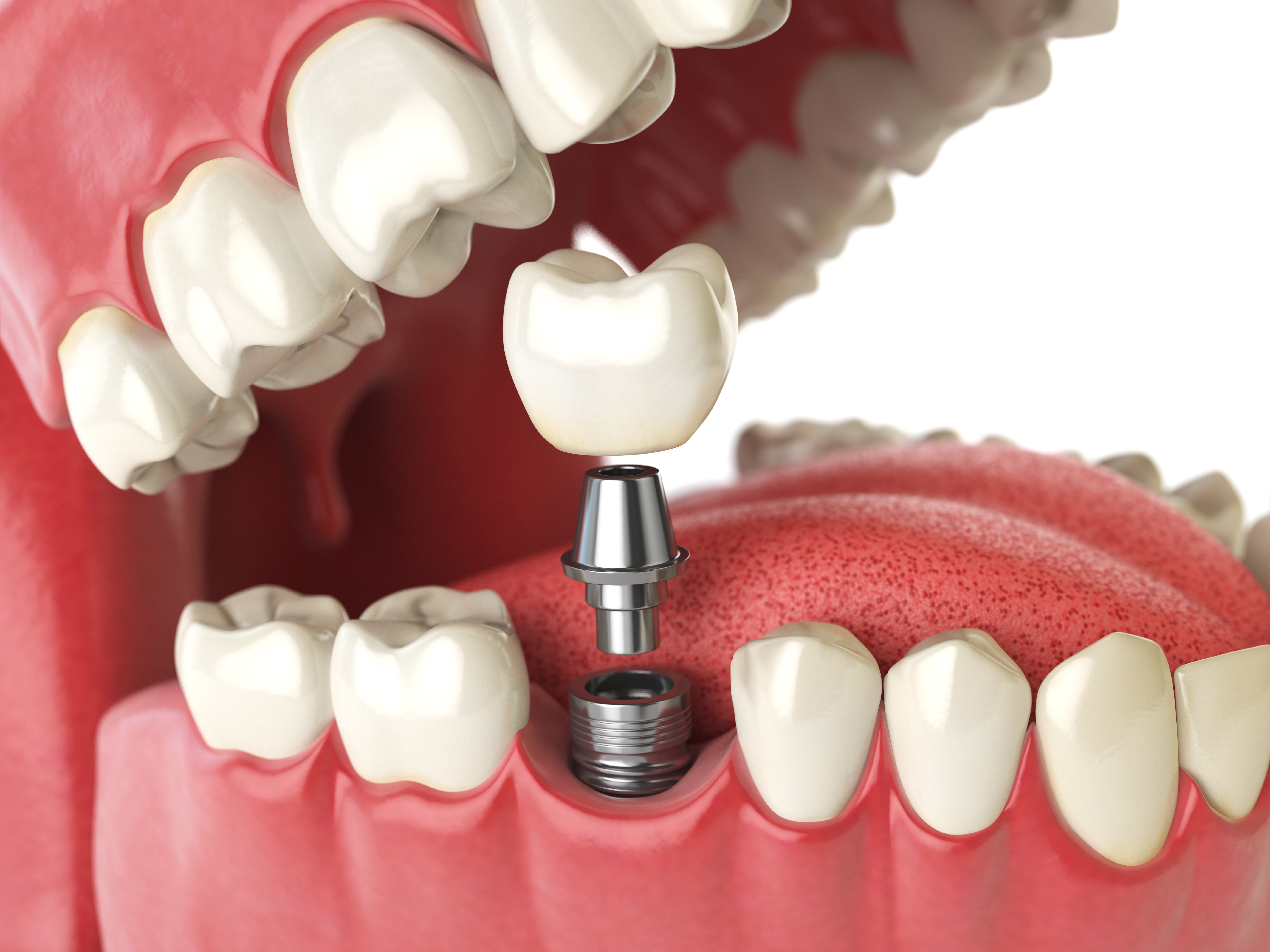
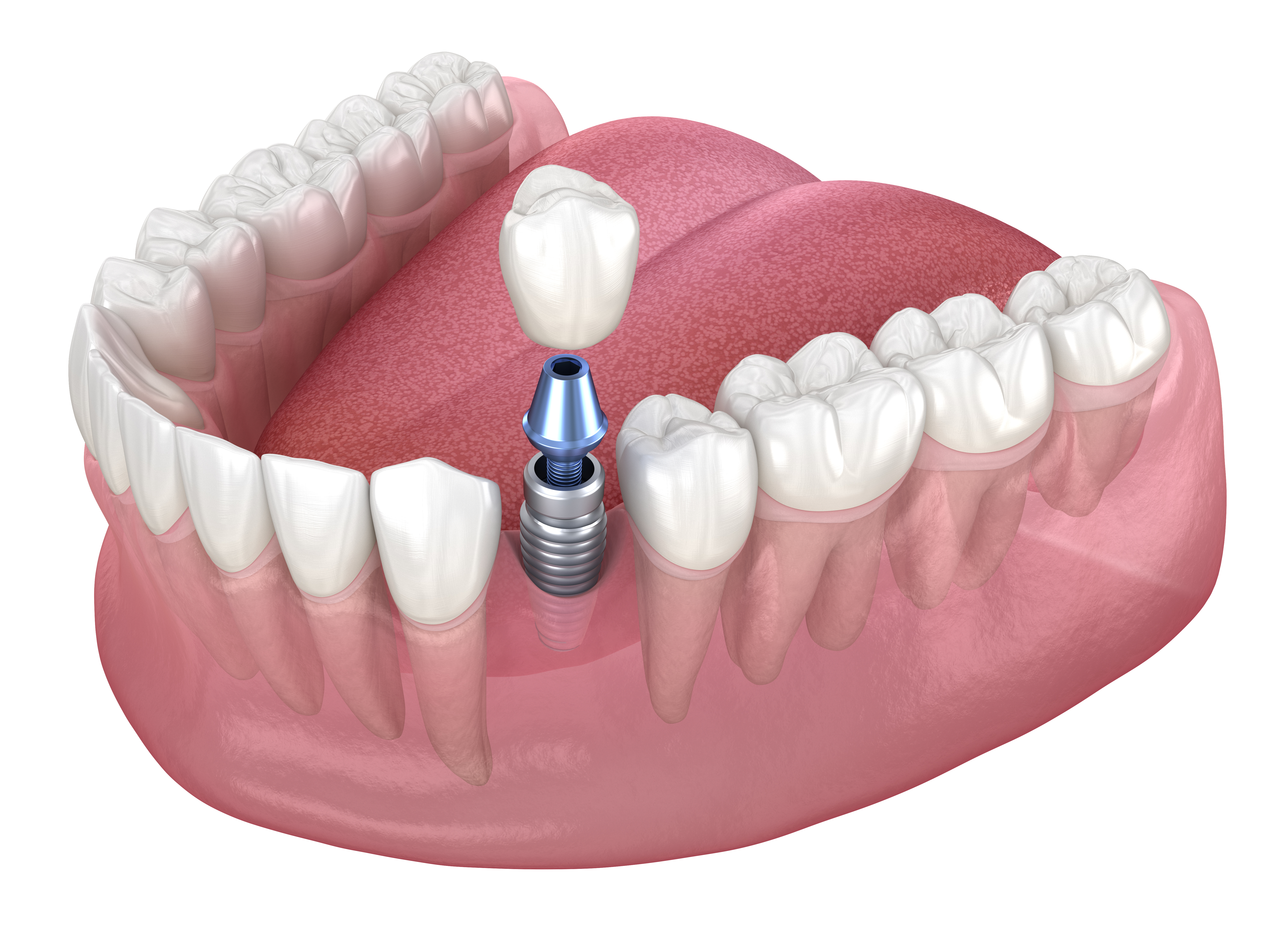
Bioreactor applications: from cell therapy to enzymes
Bioreactor applications span microbial production, mammalian cell culture, plant cell systems, and emerging modalities like cultivated meat and certain cell-based therapies. Each application reshapes what “good performance” means. In microbial enzyme production, the main goal may be volumetric productivity and consistent protein quality, with a heavy focus on feeding and aeration. In monoclonal antibody production, maintaining cell viability, controlling glycosylation-related conditions, and ensuring reproducible product quality attributes become central.
For adherent cell applications, microcarriers or fixed-bed bioreactors can increase surface area, but they add complexity in mixing and harvest. For high-density perfusion processes, cell retention devices and continuous media exchange enable stable productivity but require tight monitoring of cell density, metabolite accumulation, and filter performance.
Across applications, sterility assurance remains non-negotiable. Clean-in-place and steam-in-place routines, sterile connectors, and validated filter integrity tests reduce contamination risk. Just as important is defining critical process parameters and linking them to critical quality attributes so that process control aligns with the final product’s requirements.
Advanced techniques increasingly rely on a quality-by-design mindset: mapping how changes in agitation, feed composition, or gas strategy influence not only growth but also product variants. Digital twins and mechanistic models are used in some settings to test “what-if” scenarios, though they only add value when grounded in high-quality data and realistic assumptions.
In practice, successful bioreactor operation is the intersection of biology, fluid dynamics, measurement science, and disciplined execution. When those elements are aligned, scale-up becomes less about trial and error and more about translating a well-understood process into a larger, more demanding environment.